We use cookies to understand how you use our site and to improve your experience. This includes personalizing content and advertising. By continuing to use our site, you accept our use of cookies. View our Privacy Statement.
Share
Sterile Reprocessing Cycle
27.05.2021 • Products
Sterile Reprocessing Cycle
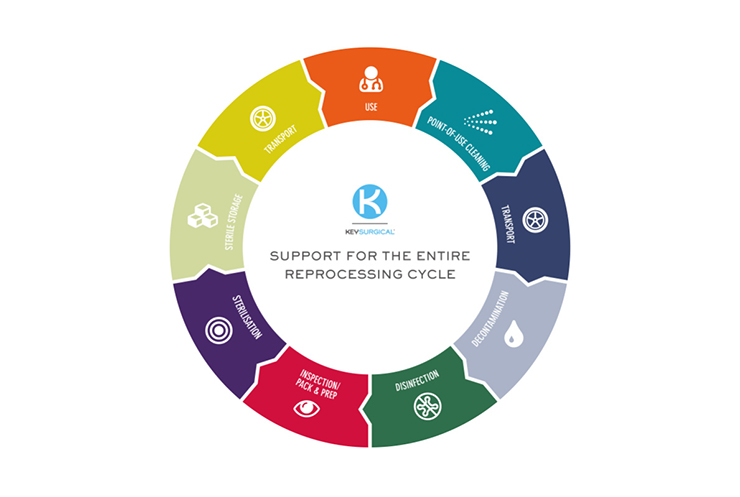
At Key Surgical we pride ourselves on offering support and solutions for the entire reprocessing cycle. Patient safety is key in everything that we do which includes to help fight bacteria or contamination on all Reusable Invasive Medical Devices (RIMD). We understand that everything that encounters the patient or within the procedure field much be systematically decontaminated after each surgical procedure. Attention much be given to all potential sources of contamination.All IFU’s that come with any device will contain the correct information to guide you if first the product is reusable to need to be decontaminated. Secondly how it needs to be contamination and the differentiates between the critical and less critical areas of the product.
As well as our extensive product range, globally and locally to support your reprocessing units, we offer best-in-class clinical support services including training for your staff. We offer full CDU reprocessing training that includes explaining all aspects of the central decontamination unit functions including cleaning, disinfection, and sterilisation. As well what causes and spreads infection and why do we need decontamination. We believe this training is just as valuable as the products themselves, all part of the service to our customers at no additional charge – we like to call that value added!
We also offer 180 and 360 audits which can either cover certain areas of the sterile store or the entire process in the decontamination room, clean room, sterile store, and transportation. We will analyse the results with you and work together to find any solutions to any problems or provide any suggestions to improve your processes. Again, this is an added extra service free of charge as we want to make sure we are bringing value to our Customers and their decontamination units.
If you would like to see our full Sterile Processing range download our new Sterile Processing or contact our brilliant Clinical team on clinical@keysurgical.co.uk
If you would like to hear about the latest news, products and educational updates sign up to our mailing list.